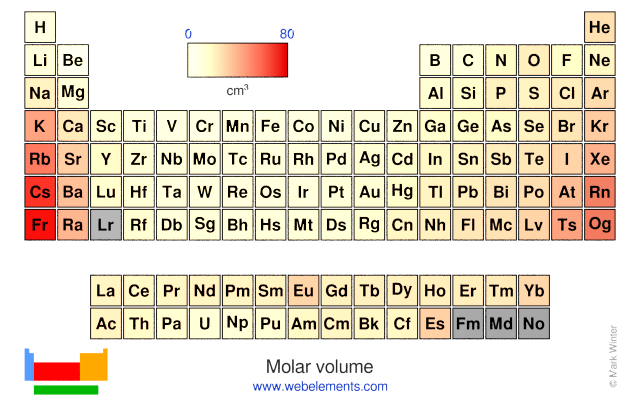
Adding an electron to an element with 3 electrons in p orbitals means the new electron must form a pair in one of the subshells -a high-energy proposition. The electron affinity for an element with a full s shell is low, since the new electron would need to be in the higher-energy p orbital. As we move down a single column on the table, the electron configuration of the outer or valence electrons remains the same, so the electron affinity and ionization energies are nearly level for lithium and sodium, for berillium and magnesium, and so on. As with ionization energy, we actually measure electron affinity as the energy required to remove the extra electron.Īgain, these are derived from experiment but trends can be predicted by looking at the periodic table. Electron affinity is the ability to attach an electron. Ionization energy is the energy required to remove an electron from the atom in the gas phase (in order to make comparisons, we need to use the same physical conditions). The increase in energy level betokens an increase in distance from the nucleus-so potassium atoms are bigger than sodium atoms. As we move from sodium (Na) to potassium (K) for example, we are moving from a 3s to a 4s orbital. Moving down the table is a different story. The increasing postive charge in the nucleus results in an increasing effective nuclear charge Z which holds the electrons closer and closer. But we have to remember that as we move from left to right, the electrons are added to the same energy level or to lower energy levels (as the d and f shells fill up), closer to the nucleus. This may seem counter intuitive-after all, each new element to the right has more electrons, which should take up more room. In general, atomic radii decrease from left to right across the periodic table. If you want to compare them to something, they are more like the gas planets Jupiter and Saturn, where gas density gradually increases as we move to the center of the planet, but where there is no planetary surface as there is on earth or Venus. These are estimates-the electron shells have no solid surfaces. How does the radius of the atom when potassium K loses an electron? When carbon loses an electron? Is there a trend in the radius change? Notice that we assume no vacuum between the atoms.Ĭompare the size of neutral atoms (red) and ions (green) in this periodic table arrangement of radius information.
For example, in diamond, the carbon nuclei are 154 picometers apart, so the diameter of each carbon atom is estimated to be half that, or 77 pm. Sizes can be estimated by finding the distance between centers of atoms in pure substances of the element (using electron microscopes to examine the surface of the sample). So mass should increase across periodic table rows from left to right and down periodic table columns experimental evidence supports this prediction.Ītomic size does not follow such simple rules. We know that mass increases as the atomic number increases, and depends primarily on the number of protons and neutrons in the atoms. We've discussed mass at length by doing molar mass conversions.
:max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
Atomic propertiesĪtomic properties include size and mass. Now we look at other "trends" in the table for atomic size, ionization energy, electron affinity, ion sizes, and chemical reaction properties.
Last time, we looked at how the periodic table can give us clues to the electron configuration of different elements in their ground state.
